| |||
| |||
| Names | |||
|---|---|---|---|
| IUPAC name
oxonium
| |||
| Other names
hydronium ion
| |||
| Identifiers | |||
3D model (JSmol)
|
|||
| ChEBI | |||
| ChemSpider | |||
| 141 | |||
PubChem CID
|
|||
CompTox Dashboard (EPA)
|
|||
| |||
| |||
| Properties | |||
| H3O+ | |||
| Molar mass | 19.023 g·mol−1 | ||
| Acidity (pKa) | 0 | ||
| Conjugate base | Water | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
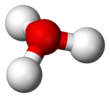
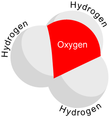
In chemistry, hydronium (hydroxonium in traditional British English) is the cation [H3O]+, also written as H3O+, the type of oxonium ion produced by protonation of water. It is often viewed as the positive ion present when an Arrhenius acid is dissolved in water, as Arrhenius acid molecules in solution give up a proton (a positive hydrogen ion, H+) to the surrounding water molecules (H2O). In fact, acids must be surrounded by more than a single water molecule in order to ionize, yielding aqueous H+ and conjugate base.
Three main structures for the aqueous proton have garnered experimental support:
- the Eigen cation, which is a tetrahydrate, H3O+(H2O)3
- the Zundel cation, which is a symmetric dihydrate, H+(H2O)2
- and the Stoyanov cation, an expanded Zundel cation, which is a hexahydrate: H+(H2O)2(H2O)4[1][2]
Spectroscopic evidence from well-defined IR spectra overwhelmingly supports the Stoyanov cation as the predominant form.[3][4][5][6][non-primary source needed] For this reason, it has been suggested that wherever possible, the symbol H+(aq) should be used instead of the hydronium ion.[2]
- ^ Reed, C.A. (2013). "Myths about the proton. The nature of H+ in condensed media". Acc. Chem. Res. 46 (11): 2567–2575. doi:10.1021/ar400064q. PMC 3833890. PMID 23875729.
- ^ a b Silverstein, Todd P. (2014). "The aqueous proton is hydrated by more than one water molecule: Is the hydronium ion a useful conceit?". J. Chem. Educ. 91 (4): 608–610. Bibcode:2014JChEd..91..608S. doi:10.1021/ed400559t.
- ^ Thamer, M.; DeMarco, L.; Ramesha, K.; Mandel, A.; Tokmakoff, A. (2015). "Ultrafast 2D IR spectroscopy of the excess proton in liquid water". Science. 350 (6256): 78–82. Bibcode:2015Sci...350...78T. doi:10.1126/science.aab3908. PMID 26430117. S2CID 27074374.
- ^ Daly Jr., C.A.; Streacker, L.M.; Sun, Y.; Pattenaude, S.R.; Hassanali, A.A.; Petersen, P.B.; et al. (2017). "Decomposition of the experimental Raman and IR spectra of acidic water into proton, special pair, and counterion contributions". J. Phys. Chem. Lett. 8 (21): 5246–5252. doi:10.1021/acs.jpclett.7b02435. PMID 28976760.
- ^ Dahms, F.; Fingerhut, B.P.; Nibbering, E.T.; Pines, E.; Elsaesser, T. (2017). "Large-amplitude transfer motion of hydrated excess protons mapped by ultrafast 2D IR spectroscopy". Science. 357 (6350): 491–495. Bibcode:2017Sci...357..491D. doi:10.1126/science.aan5144. PMID 28705988. S2CID 40492001.
- ^ Fournier, J.A.; Carpenter, W.B.; Lewis, N.H.; Tokmakoff, A. (2018). "Broadband 2D IR spectroscopy reveals dominant asymmetric H5O2+ proton hydration structures in acid solutions". Nature Chemistry. 10 (9): 932–937. Bibcode:2018NatCh..10..932F. doi:10.1038/s41557-018-0091-y. OSTI 1480907. PMID 30061612. S2CID 51882732.