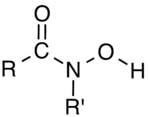
In organic chemistry, hydroxamic acids are a class of organic compounds having a general formula R−C(=O)−N(−OH)−R' bearing the functional group −C(=O)−N(−OH)−, where R and R' are typically organyl groups (e.g., alkyl or aryl) or hydrogen. They are amides (R−C(=O)−NH−R') wherein the nitrogen atom has a hydroxyl (−OH) substituent. They are often used as metal chelators.[1]
Common example of hydroxamic acid is aceto-N-methylhydroxamic acid (H3C−C(=O)−N(−OH)−CH3). Some uncommon examples of hydroxamic acids are formo-N-chlorohydroxamic acid (H−C(=O)−N(−OH)−Cl) and chloroformo-N-methylhydroxamic acid (Cl−C(=O)−N(−OH)−CH3).
- ^ Munson, James W. (1992). "Hydroxamic acids". In S. Patai (ed.). Acid Derivatives (1992), Volume 2. PATAI'S Chemistry of Functional Groups. pp. 849–873. doi:10.1002/9780470772508.ch15. ISBN 9780470772508.