 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Droxia, Hydrea, Siklos, others |
| Other names | Hydroxyurea (USAN US) |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a682004 |
| License data |
|
| Pregnancy category |
|
| Routes of administration | By mouth |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Metabolism | Liver (to CO2 and urea) |
| Elimination half-life | 2–4 hours |
| Excretion | Kidney and lungs |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| NIAID ChemDB | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.004.384 |
| Chemical and physical data | |
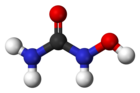
| Formula | CH4N2O2 |
| Molar mass | 76.055 g·mol−1 |
| 3D model (JSmol) | |
| Melting point | 133 to 136 °C (271 to 277 °F) |
| |
| |
| (verify) | |
Hydroxycarbamide, also known as hydroxyurea, is a medication used in sickle-cell disease, essential thrombocythemia, chronic myelogenous leukemia, polycythemia vera, and cervical cancer.[4][5] In sickle-cell disease it increases fetal hemoglobin and decreases the number of attacks.[4] It is taken by mouth.[4]
Common side effects include bone marrow suppression, fevers, loss of appetite, psychiatric problems, shortness of breath, and headaches.[4][5] There is also concern that it increases the risk of later cancers.[4] Use during pregnancy is typically harmful to the fetus.[4] Hydroxycarbamide is in the antineoplastic family of medications. It is believed to work by blocking the making of DNA.[4]
Hydroxycarbamide was approved for medical use in the United States in 1967.[4] It is on the World Health Organization's List of Essential Medicines.[6] Hydroxycarbamide is available as a generic medication.[4]
- ^ "FDA-sourced list of all drugs with black box warnings (Use Download Full Results and View Query links.)". nctr-crs.fda.gov. FDA. Retrieved 22 October 2023.
- ^ "Xromi- hydroxyurea solution". DailyMed. 8 April 2024. Retrieved 18 May 2024.
- ^ "Siklos EPAR". European Medicines Agency. 9 July 2003. Retrieved 24 July 2024.
- ^ a b c d e f g h i "Hydroxyurea". The American Society of Health-System Pharmacists. Retrieved 8 December 2016.
- ^ a b "Hydrea 500 mg Hard Capsules – Summary of Product Characteristics (SPC) – (eMC)". www.medicines.org.uk. Archived from the original on 20 December 2016. Retrieved 14 December 2016.
- ^ World Health Organization (2019). World Health Organization model list of essential medicines: 21st list 2019. Geneva: World Health Organization. hdl:10665/325771. WHO/MVP/EMP/IAU/2019.06. License: CC BY-NC-SA 3.0 IGO.