 | |
| Clinical data | |
|---|---|
| Trade names | Ketas, Pinatos, Eyevinal |
| AHFS/Drugs.com | International Drug Names |
| Routes of administration | By mouth (capsules), topical (ophthalmic solution) |
| ATC code | |
| Legal status | |
| Legal status | |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.164.881 |
| Chemical and physical data | |
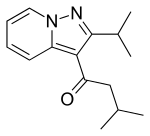
| Formula | C14H18N2O |
| Molar mass | 230.311 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
Ibudilast (development codes: AV-411 or MN-166) is an anti-inflammatory drug used mainly in Japan, which acts as a phosphodiesterase inhibitor, inhibiting the PDE4 subtype to the greatest extent,[1] but also showing significant inhibition of other PDE subtypes.[2][3]
- ^ Huang Z, Liu S, Zhang L, Salem M, Greig GM, Chan CC, et al. (May 2006). "Preferential inhibition of human phosphodiesterase 4 by ibudilast". Life Sciences. 78 (23): 2663–2668. doi:10.1016/j.lfs.2005.10.026. PMID 16313925.
- ^ Suzumura A, Ito A, Yoshikawa M, Sawada M (August 1999). "Ibudilast suppresses TNFalpha production by glial cells functioning mainly as type III phosphodiesterase inhibitor in the CNS". Brain Research. 837 (1–2): 203–212. doi:10.1016/s0006-8993(99)01666-2. PMID 10434004. S2CID 7910607.
- ^ Gibson LC, Hastings SF, McPhee I, Clayton RA, Darroch CE, Mackenzie A, et al. (May 2006). "The inhibitory profile of Ibudilast against the human phosphodiesterase enzyme family". European Journal of Pharmacology. 538 (1–3): 39–42. doi:10.1016/j.ejphar.2006.02.053. PMID 16674936.