This article needs attention from an expert in Chemicals. The specific problem is: Is iron (III) acetate actually soluble or not?. See the talk page for details. (August 2024) |
| |
| |
| Names | |
|---|---|
| IUPAC name
iron(III) acetate
| |
| Other names
basic iron(III) acetate , iron(III) oxyacetate, iron(III) Acetate
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
PubChem CID
|
|
| UNII | |
| |
| |
| Properties | |
| C14H27Fe3O18 | |
| Molar mass | 650.9 g/mol |
| Appearance | brownish-red powder |
| Insoluble | |
| Solubility | soluble in ethanol[2] |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
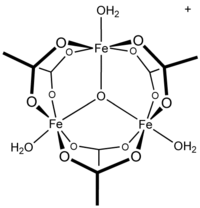
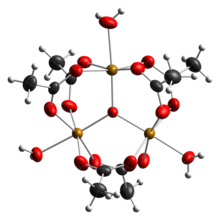
Ferric acetate is the acetate salt of the coordination complex [Fe3O(OAc)6(H2O)3]+ (OAc− is CH3CO2−). Commonly the salt is known as "basic iron acetate".[3] The formation of the red-brown complex was once used as a test for ferric ions.[4]
- ^ Lide, David R., ed. (2006). CRC Handbook of Chemistry and Physics (87th ed.). Boca Raton, FL: CRC Press. pp. 4–63. ISBN 0-8493-0487-3.
- ^ "Iron(III) Acetate". EndMemo. Retrieved 18 April 2015.
- ^ J., Burgess; M. V., Twigg (2005). R. Bruce, King; J., Wiley (eds.). Encyclopedia of inorganic chemistry (2nd ed.). New York: Wiley. ISBN 978-0-470-86078-6.
- ^ H., Brearley; F., Ibbotson (1902). The Analysis of Steel-Works Materials. London ; New York: Longmans, Green. Archived from the original on 18 April 2015.