 | |
| Clinical data | |
|---|---|
| Trade names | Sporanox, Sporaz, Orungal, others |
| Other names | ITZ |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a692049 |
| License data |
|
| Pregnancy category |
|
| Routes of administration | By mouth, solution), vaginal suppository, intravenous |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | ~55%, maximal if taken with full meal |
| Protein binding | 99.8% |
| Metabolism | Extensive in liver (CYP3A4) |
| Metabolites | Hydroxy-itraconazole, keto-itraconazole, N-desalkyl-itraconazole[4] |
| Elimination half-life | 21 hours |
| Excretion | Kidney (35%), faeces (54%)[5] |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.123.596 |
| Chemical and physical data | |
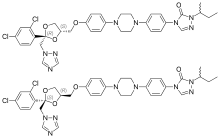
| Formula | C35H38Cl2N8O4 |
| Molar mass | 705.64 g·mol−1 |
| 3D model (JSmol) | |
| Chirality | Racemic mixture |
| Melting point | 165[6] °C (329 °F) |
| Solubility in water | 7.8 ± 0.4 × 10−6 mol/L (pH 1.6)[6] mg/mL (20 °C) |
| |
| |
| (verify) | |
Itraconazole, sometimes abbreviated ITZ, is an antifungal medication used to treat a number of fungal infections.[7] This includes aspergillosis, blastomycosis, coccidioidomycosis, histoplasmosis, and paracoccidioidomycosis.[7] It may be given by mouth or intravenously.[7]
Common side effects include nausea, diarrhea, abdominal pain, rash, and headache.[7] Severe side effects may include liver problems, heart failure, Stevens–Johnson syndrome and allergic reactions including anaphylaxis.[7] It is unclear if use during pregnancy or breastfeeding is safe.[1] It is in the triazole family of medications.[7] It stops fungal growth by affecting the cell membrane or affecting their metabolism.[7]
Itraconazole was patented in 1978 and approved for medical use in the United States in 1992.[7][8] It is on the World Health Organization's List of Essential Medicines.[9]
Recent research works suggest itraconazole (ITZ) could also be used in the treatment of cancer by inhibiting the hedgehog pathway[10] in a similar way to sonidegib.
- ^ a b "Itraconazole Use During Pregnancy". Drugs.com. 20 March 2019. Retrieved 15 May 2020.
- ^ "FDA-sourced list of all drugs with black box warnings (Use Download Full Results and View Query links.)". nctr-crs.fda.gov. FDA. Retrieved 22 October 2023.
- ^ "Sporanox 10 mg/mL Oral Solution - Summary of Product Characteristics (SmPC)". (emc). 1 February 2018. Retrieved 15 May 2020.
- ^ Isoherranen N, Kunze KL, Allen KE, Nelson WL, Thummel KE (October 2004). "Role of itraconazole metabolites in CYP3A4 inhibition". Drug Metabolism and Disposition. 32 (10): 1121–1131. doi:10.1124/dmd.104.000315. PMID 15242978. S2CID 6941636.
- ^ "Sporanox (itraconazole) Capsules. Full Prescribing Information" (PDF). Janssen Pharmaceuticals, Inc. Archived from the original (PDF) on 17 May 2018. Retrieved 28 August 2016.
- ^ a b Vasilev NA, Surov AO, Voronin AP, Drozd KV, Perlovich GL (April 2021). "Novel cocrystals of itraconazole: Insights from phase diagrams, formation thermodynamics and solubility". International Journal of Pharmaceutics. 599: 120441. doi:10.1016/j.ijpharm.2021.120441. PMID 33675927. S2CID 232135660.
- ^ a b c d e f g h "Itraconazole". The American Society of Health-System Pharmacists. Retrieved 8 December 2017.
- ^ Fischer J, Ganellin CR (2006). Analogue-based Drug Discovery. John Wiley & Sons. p. 503. ISBN 9783527607495.
- ^ World Health Organization (2019). World Health Organization model list of essential medicines: 21st list 2019. Geneva: World Health Organization. hdl:10665/325771. WHO/MVP/EMP/IAU/2019.06. License: CC BY-NC-SA 3.0 IGO.
- ^ Li K, Fang D, Xiong Z, Luo R (2019). "Inhibition of the hedgehog pathway for the treatment of cancer using Itraconazole". OncoTargets and Therapy. 12: 6875–6886. doi:10.2147/OTT.S223119. PMC 6711563. PMID 31692536.