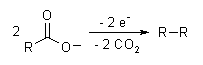The Kolbe electrolysis or Kolbe reaction is an organic reaction named after Hermann Kolbe.[1] The Kolbe reaction is formally a decarboxylative dimerisation of two carboxylic acids (or carboxylate ions). The overall reaction is:
If a mixture of two different carboxylates are used, all combinations of them are generally seen as the organic product structures:
- 3 R1COO− + 3 R2COO− → R1−R1 + R1−R2 + R2−R2 + 6 CO2 + 6 e−
The reaction mechanism involves a two-stage radical process: electrochemical decarboxylation gives a radical intermediate, which combine to form a covalent bond.[2] As an example, electrolysis of acetic acid yields ethane and carbon dioxide:
- CH3COOH → CH3COO− → CH3COO· → CH3· + CO2
- 2CH3· → CH3CH3
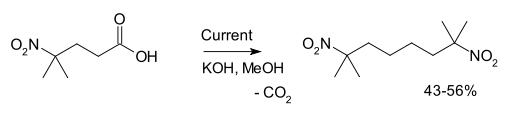
Another example is the synthesis of 2,7-dimethyl-2,7-dinitrooctane from 4-methyl-4-nitrovaleric acid:[3]
The Kolbe reaction has also been occasionally used in cross-coupling reactions.
In 2022, it was discovered that the Kolbe electrolysis is enhanced if an alternating square wave current is used instead of a direct current.[4][5]
- ^ Utley, James (1997). "Trends in Organic Electrosynthesis". Chemical Society Reviews. 26 (3): 157. doi:10.1039/cs9972600157.
- ^ Vijh, A. K.; Conway, B. E. (1967). "Electrode Kinetic Aspects of the Kolbe Reaction". Chem Rev. 67 (6): 623–664. doi:10.1021/cr60250a003.
- ^ Sharkey, W. H.; Langkammerer, C. M. (1973). "2,7-Dimethyl-2,7-dinitrooctane". Organic Syntheses; Collected Volumes, vol. 5, p. 445.
- ^ Hioki, Yuta; Costantini, Matteo; Griffin, Jeremy; Harper, Kaid; Prado Merini, Melania; Nissl, Benedikt; Kawamata, Yu; Baran, Phil (31 October 2022). Overcoming the Limitations of Kolbe Coupling via Waveform-Controlled Electrosynthesis (Report). Chemistry. doi:10.26434/chemrxiv-2022-3cj82-v2.
- ^ Hioki, Yuta; Costantini, Matteo; Griffin, Jeremy; Harper, Kaid C.; Merini, Melania Prado; Nissl, Benedikt; Kawamata, Yu; Baran, Phil S. (7 April 2023). "Overcoming the limitations of Kolbe coupling with waveform-controlled electrosynthesis". Science. 380 (6640): 81–87. doi:10.1126/science.adf4762. ISSN 0036-8075. PMID 37023204.