 __ La3+ __ OH−
| |
| Names | |
|---|---|
| IUPAC name
Lanthanum(III) hydroxide
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
| ECHA InfoCard | 100.034.994 |
| EC Number |
|
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
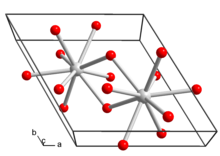
| La(OH)3 | |
| Molar mass | 189.93 g/mol |
| Ksp= 2.00·10−21 | |
| Structure | |
| hexagonal | |
| P63/m, No. 176 | |
a = 6.547 Å, c = 3.854 Å
| |
| Hazards | |
| Occupational safety and health (OHS/OSH): | |
Main hazards
|
Irritant |
| GHS labelling:[1] | |

| |
| Danger | |
| H314 | |
| P260, P264, P280, P301+P330+P331, P303+P361+P353, P304+P340, P305+P351+P338, P310, P321, P363, P405, P501 | |
| NFPA 704 (fire diamond) | |
| Flash point | Non-flammable |
| Related compounds | |
Other anions
|
Lanthanum(III) chloride |
Other cations
|
Cerium(III) hydroxide Actinium(III) hydroxide |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Lanthanum hydroxide is La(OH)
3, a hydroxide of the rare-earth element lanthanum.
- ^ "C&L Inventory". echa.europa.eu.
