 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Xalatan, others |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a697003 |
| License data |
|
| Pregnancy category |
|
| Routes of administration | Topical eye drop |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Metabolism | Activation by ester hydrolysis, deactivation by beta oxidation |
| Onset of action | 3–4 hours |
| Elimination half-life | 17 minutes (plasma) |
| Duration of action | ≥ 24 hours |
| Excretion | Mainly via kidney |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.162.178 |
| Chemical and physical data | |
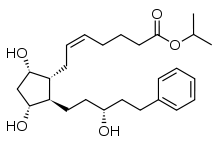
| Formula | C26H40O5 |
| Molar mass | 432.601 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| (verify) | |
Latanoprost, sold under the brand name Xalatan among others, is a medication used to treat increased pressure inside the eye (intraocular pressure).[5] This includes ocular hypertension and open-angle glaucoma.[5] Latanaprost is applied as eye drops to the eyes.[5] Onset of effects is usually within four hours, and they last for up to a day.[5]
Common side effects include blurry vision, redness of the eye, itchiness, and darkening of the iris.[5] Latanoprost is in the prostaglandin analogue family of medications.[5] It works by increasing the outflow of aqueous fluid from the eyes through the uveoscleral tract.[6]
Latanoprost was approved for medical use in the United States and the European Union in 1996.[5][3] It is on the World Health Organization's List of Essential Medicines.[7] Latanoprost is available as a generic medication.[8] In 2022, it was the 67th most commonly prescribed medication in the United States, with more than 9 million prescriptions.[9][10] It is available as a fixed-dose combination with netarsudil as netarsudil/latanoprost and with timolol as latanoprost/timolol.
- ^ "Latanoprost 50 micrograms/ml eye drops, solution - Summary of Product Characteristics (SmPC)". (emc). 1 July 2022. Archived from the original on 1 July 2022. Retrieved 1 July 2022.
- ^ Cite error: The named reference
Xalatan FDA labelwas invoked but never defined (see the help page). - ^ a b "Catiolanze EPAR". European Medicines Agency (EMA). 14 September 2023. Archived from the original on 5 December 2023. Retrieved 11 December 2023.
- ^ "Catiolanze Product information". Union Register of medicinal products. 16 November 2023. Retrieved 11 December 2023.
- ^ a b c d e f g "Latanoprost Monograph". The American Society of Health-System Pharmacists. Archived from the original on 28 December 2016. Retrieved 8 December 2016.
- ^ Patel SS, Spencer CM (1996). "Latanoprost. A review of its pharmacological properties, clinical efficacy and tolerability in the management of primary open-angle glaucoma and ocular hypertension". Drugs Aging. 9 (5): 363–378. doi:10.2165/00002512-199609050-00007. PMID 8922563. S2CID 25169085.
- ^ World Health Organization (2023). The selection and use of essential medicines 2023: web annex A: World Health Organization model list of essential medicines: 23rd list (2023). Geneva: World Health Organization. hdl:10665/371090. WHO/MHP/HPS/EML/2023.02.
- ^ Hamilton R (2015). Tarascon Pocket Pharmacopoeia 2015 Deluxe Lab-Coat Edition. Jones & Bartlett Learning. p. 413. ISBN 9781284057560.
- ^ "The Top 300 of 2022". ClinCalc. Archived from the original on 30 August 2024. Retrieved 30 August 2024.
- ^ "Latanoprost Drug Usage Statistics, United States, 2013 - 2022". ClinCalc. Retrieved 30 August 2024.