 | |
| Clinical data | |
|---|---|
| Trade names | Sunlenca |
| Other names | GS-CA1, GS-6207 |
| License data |
|
| Pregnancy category |
|
| Routes of administration | By mouth, subcutaneous |
| Drug class | Capsid inhibitors |
| ATC code | |
| Legal status | |
| Legal status | |
| Identifiers | |
| |
| CAS Number | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| PDB ligand | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
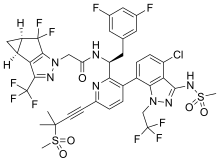
| Formula | C39H32ClF10N7O5S2 |
| Molar mass | 968.28 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Lenacapavir, sold under the brand name Sunlenca, is an antiretroviral medication used to treat HIV/AIDS.[9][10] It is taken by mouth or by subcutaneous injection.[9][10]
The most common side effects include reactions at the injection site and nausea.[10][11]
Lenacapavir was approved for medical treatment in the European Union in August 2022,[10][12] in Canada in November 2022,[5][6] and in the United States in December 2022.[9][11][13][14] It is the first of a class of drugs called capsid inhibitors to be approved by the US Food and Drug Administration (FDA) for treating HIV/AIDS.[11][15]
- ^ a b "Sunlenca". Therapeutic Goods Administration. 6 April 2023. Archived from the original on 8 April 2023. Retrieved 7 April 2023.
- ^ "Sunlenca lenacapavir (as sodium) 300 mg film coated tablet blister pack (392350)". Therapeutic Goods Administration. 28 March 2023. Archived from the original on 8 April 2023. Retrieved 7 April 2023.
- ^ "Sunlenca lenacapavir (as sodium) 463.5 mg/1.5 mL solution for injection vial (386895)". Therapeutic Goods Administration. 28 March 2023. Archived from the original on 8 April 2023. Retrieved 7 April 2023.
- ^ https://www.tga.gov.au/resources/auspar/auspar-sunlenca [bare URL]
- ^ a b "Sunlenca Oral Product information". Health Canada. 25 April 2012. Archived from the original on 15 January 2023. Retrieved 23 December 2022.
- ^ a b "Sunlenca Subcutaneous Product information". Health Canada. 25 April 2012. Archived from the original on 15 January 2023. Retrieved 23 December 2022.
- ^ "Summary Basis of Decision - Sunlenca". Health Canada. 10 March 2023. Archived from the original on 25 April 2023. Retrieved 24 April 2023.
- ^ "Details for: Sunlenca". Health Canada. 15 March 2023. Archived from the original on 3 March 2024. Retrieved 3 March 2024.
- ^ a b c d "Sunlenca- lenacapavir sodium tablet, film coated Sunlenca- lenacapavir sodium kit". DailyMed. 21 December 2022. Archived from the original on 21 January 2023. Retrieved 21 January 2023.
- ^ a b c d e "Sunlenca EPAR". European Medicines Agency (EMA). 22 June 2022. Archived from the original on 26 August 2022. Retrieved 25 August 2022. Text was copied from this source which is copyright European Medicines Agency. Reproduction is authorized provided the source is acknowledged.
- ^ a b c "FDA Approves New HIV Drug for Adults with Limited Treatment Options" (Press release). U.S. Food and Drug Administration (FDA). 22 December 2022. Archived from the original on 15 January 2023. Retrieved 23 December 2022.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain.
- ^ "Gilead Announces First Global Regulatory Approval of Sunlenca (Lenacapavir), the Only Twice-Yearly HIV Treatment Option". Gilead Sciences, Inc. (Press release). 22 August 2022. Archived from the original on 15 January 2023. Retrieved 23 December 2022.
- ^ "Sunlenca (lenacapavir) Receives FDA Approval as a First-in-Class, Twice-Yearly Treatment Option for People Living With Multi-Drug Resistant HIV". Gilead Sciences, Inc. (Press release). 22 December 2022. Archived from the original on 23 December 2022. Retrieved 23 December 2022.
- ^ Paik J (September 2022). "Lenacapavir: First Approval". Drugs. 82 (14): 1499–1504. doi:10.1007/s40265-022-01786-0. PMC 10267266. PMID 36272024.
- ^ Cite error: The named reference
New Drug Therapy Approvals 2022was invoked but never defined (see the help page).