Middle: Cocaine with its numerical substitution position locants.
2′ (6′) = ortho, 3′ (5′) = meta & 4′ = para
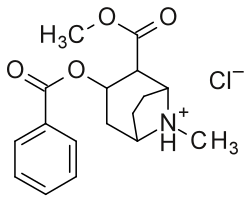
Bottom: Alternate two-dimensional molecular diagram of cocaine; shown specifically as a protonated, NH+, hydrochloride, and disregarding 3D stereochemistry
This is a list of cocaine analogues. A cocaine analogue is an (usually) artificial construct of a novel chemical compound from (often the starting point of natural) cocaine's molecular structure, with the result product sufficiently similar to cocaine to display similarity in, but alteration to, its chemical function. Within the scope of analogous compounds created from the structure of cocaine, so named "cocaine analogues" retain 3β-benzoyloxy or similar functionality (the term specifically used usually distinguishes from phenyltropanes, but in the broad sense generally, as a category, includes them) on a tropane skeleton, as compared to other stimulants of the kind. Many of the semi-synthetic cocaine analogues proper which have been made & studied have consisted of among the nine following classes of compounds:[a]
- stereoisomers of cocaine
- 3β-phenyl ring substituted analogues
- 2β-substituted analogues
- N-modified analogues of cocaine
- 3β-carbamoyl analogues
- 3β-alkyl-3-benzyl tropanes
- 6/7-substituted cocaines
- 6-alkyl-3-benzyl tropanes
- piperidine homologues of cocaine
However strict analogues of cocaine would also include such other potential combinations as phenacyltropanes & other carbon branched replacements not listed above. The term may also be loosely used to refer to drugs manufactured from cocaine or having their basis as a total synthesis of cocaine, but modified to alter their effect & QSAR. These include both intracellular sodium channel blocker anaesthetics and stimulant dopamine reuptake inhibitor ligands (such as certain, namely tropane-bridged-excised, piperidines). Additionally, researchers have supported combinatorial approaches for taking the most promising analogues currently elucidated and mixing them to the end of discovering novel & efficacious compounds to optimize their utilization for differing distinct specified purposes.[b]
- ^ a b Singh, Satendra; et al. (2000). "Chemistry, Design, and Structure-Activity Relationship of Cocaine Antagonists" (PDF). Chem. Rev. 100 (3): 925–1024. doi:10.1021/cr9700538. PMID 11749256.
Cite error: There are <ref group=lower-alpha> tags or {{efn}} templates on this page, but the references will not show without a {{reflist|group=lower-alpha}} template or {{notelist}} template (see the help page).