It has been suggested that this article be merged with Macromolecular cages. (Discuss) Proposed since November 2024. |
This article includes a list of general references, but it lacks sufficient corresponding inline citations. (October 2019) |
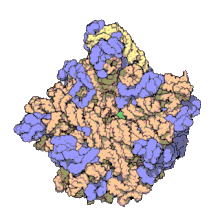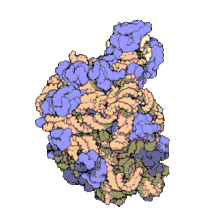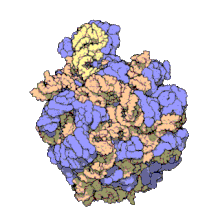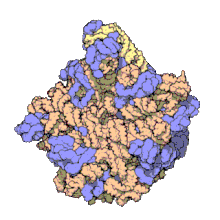
In molecular biology, the term macromolecular assembly (MA) refers to massive chemical structures such as viruses and non-biologic nanoparticles, cellular organelles and membranes and ribosomes, etc. that are complex mixtures of polypeptide, polynucleotide, polysaccharide or other polymeric macromolecules. They are generally of more than one of these types, and the mixtures are defined spatially (i.e., with regard to their chemical shape), and with regard to their underlying chemical composition and structure. Macromolecules are found in living and nonliving things, and are composed of many hundreds or thousands of atoms held together by covalent bonds; they are often characterized by repeating units (i.e., they are polymers). Assemblies of these can likewise be biologic or non-biologic, though the MA term is more commonly applied in biology, and the term supramolecular assembly is more often applied in non-biologic contexts (e.g., in supramolecular chemistry and nanotechnology). MAs of macromolecules are held in their defined forms by non-covalent intermolecular interactions (rather than covalent bonds), and can be in either non-repeating structures (e.g., as in the ribosome (image) and cell membrane architectures), or in repeating linear, circular, spiral, or other patterns (e.g., as in actin filaments and the flagellar motor, image). The process by which MAs are formed has been termed molecular self-assembly, a term especially applied in non-biologic contexts. A wide variety of physical/biophysical, chemical/biochemical, and computational methods exist for the study of MA; given the scale (molecular dimensions) of MAs, efforts to elaborate their composition and structure and discern mechanisms underlying their functions are at the forefront of modern structure science.

- ^ Ban N, Nissen P, Hansen J, Moore PB, Steitz TA (August 2000). "The complete atomic structure of the large ribosomal subunit at 2.4 A resolution". Science. 289 (5481): 905–920. Bibcode:2000Sci...289..905B. CiteSeerX 10.1.1.58.2271. doi:10.1126/science.289.5481.905. PMID 10937989.
- ^ McClure W. "50S Ribosome Subunit". Archived from the original on 2005-11-24. Retrieved 2019-10-09.
- ^ Osborne AR, Rapoport TA, van den Berg B (2005). "Protein translocation by the Sec61/SecY channel". Annual Review of Cell and Developmental Biology. 21: 529–550. doi:10.1146/annurev.cellbio.21.012704.133214. PMID 16212506.