
| |
| |
| |

| |
| Names | |
|---|---|
| Preferred IUPAC name
(2Z)-But-2-enedioic acid | |
| Other names
(Z)-Butenedioic acid; cis-Butenedioic acid; Malenic acid; Maleinic acid; Toxilic acid
| |
| Identifiers | |
3D model (JSmol)
|
|
| 3DMet | |
| 605762 | |
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| DrugBank | |
| ECHA InfoCard | 100.003.403 |
| EC Number |
|
| 49854 | |
| KEGG | |
PubChem CID
|
|
| RTECS number |
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C4H4O4 | |
| Molar mass | 116.072 g·mol−1 |
| Appearance | White solid |
| Density | 1.59 g/cm3 [1] |
| Melting point | 135 °C (275 °F; 408 K) (decomposes)[2] |
| 478.8 g/L at 20 C[2] | |
| Acidity (pKa) | pka1 = 1.90 pka2 = 6.07 [3] |
| -49.71·10−6 cm3/mol | |
| Hazards | |
| GHS labelling: | |

| |
| Warning | |
| H302, H315, H317, H319, H335 | |
| P261, P264, P270, P271, P272, P280, P301+P312, P302+P352, P304+P340, P305+P351+P338, P312, P321, P330, P332+P313, P333+P313, P337+P313, P362, P363, P403+P233, P405, P501 | |
| NFPA 704 (fire diamond) | |
| Safety data sheet (SDS) | MSDS from J. T. Baker |
| Related compounds | |
Related carboxylic acids
|
fumaric acid succinic acid crotonic acid |
Related compounds
|
maleic anhydride maleimide |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
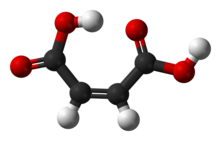
Maleic acid or cis-butenedioic acid is an organic compound that is a dicarboxylic acid, a molecule with two carboxyl groups. Its chemical formula is HO2CCH=CHCO2H. Maleic acid is the cis isomer of butenedioic acid, whereas fumaric acid is the trans isomer. Maleic acid is mainly used as a precursor to fumaric acid, and relative to its parent maleic anhydride, which has many applications.[4]
- ^ Budavari, Susan, ed. (1996), The Merck Index: An Encyclopedia of Chemicals, Drugs, and Biologicals (12th ed.), Merck, ISBN 0911910123
- ^ a b Record in the GESTIS Substance Database of the Institute for Occupational Safety and Health
- ^ CRC Handbook of Chemistry and Physics, 73rd ed.; CRC Press: Boca Raton, Florida., 1993
- ^ Cite error: The named reference
Ullmannwas invoked but never defined (see the help page).
