This article needs more reliable medical references for verification or relies too heavily on primary sources. (January 2021) |  |
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Noopept |
| Other names | omberacetam; GVS-111; DVD-111; SGS-111; benzylcarbonyl-Pro-Gly-OEt |
| Legal status | |
| Legal status |
|
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
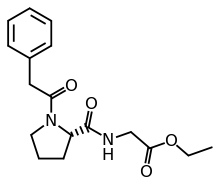
| Formula | C17H22N2O4 |
| Molar mass | 318.373 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
N-Phenylacetyl-l-prolylglycine ethyl ester is promoted as a nootropic and is a prodrug of cyclic glycine-proline.[a][2] Other names include the brand name Noopept (Russian: Ноопепт), developmental code GVS-111, and proposed INN omberacetam.[2][3][4]
Its synthesis was first reported in 1996.[2] It is orally available. As of 2017, its metabolism and elimination half-life in humans were not well understood.[2]
It has been evaluated for neuroprotective effects in treating brain injuries and stroke.[5]
- ^ "Omberacetam". Inxight. National Center for Advancing Translational Sciences (NCATS). 4QBJ98683M.
- ^ a b c d "Noopept Information". Examine.com. Retrieved 6 April 2017.
- ^ "Proposed INN List 117". WHO Drug Information. 31 (2): 308. 2017.
- ^ "Omberacetam". AdisInsight. Springer Nature Switzerland AG. Retrieved 12 May 2018.
Alternative Names: DVD-111; GVS 111; Noopept
- ^ Ostrovskaia RU, Gudasheva TA, Voronina TA, Seredenin SB (2002). "[The original novel nootropic and neuroprotective agent noopept]" [The original novel nootropic and neuroprotective agent noopept]. Eksperimental'naia i Klinicheskaia Farmakologiia [Experimental and Clinical Pharmacology] (in Russian). 65 (5): 66–72. PMID 12596521.
Cite error: There are <ref group=lower-alpha> tags or {{efn}} templates on this page, but the references will not show without a {{reflist|group=lower-alpha}} template or {{notelist}} template (see the help page).