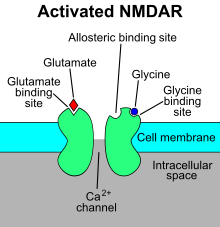
The N-methyl-D-aspartate receptor (also known as the NMDA receptor or NMDAR), is a glutamate receptor and predominantly Ca2+ ion channel found in neurons.[3][4] The NMDA receptor is one of three types of ionotropic glutamate receptors, the other two being AMPA and kainate receptors. Depending on its subunit composition, its ligands are glutamate and glycine (or D-serine). However, the binding of the ligands is typically not sufficient to open the channel as it may be blocked by Mg2+ ions which are only removed when the neuron is sufficiently depolarized. Thus, the channel acts as a "coincidence detector" and only once both of these conditions are met, the channel opens and it allows positively charged ions (cations) to flow through the cell membrane.[5] The NMDA receptor is thought to be very important for controlling synaptic plasticity and mediating learning and memory functions.[6]
The NMDA receptor is ionotropic, meaning it is a protein which allows the passage of ions through the cell membrane.[7] The NMDA receptor is so named because the agonist molecule N-methyl-D-aspartate (NMDA) binds selectively to it, and not to other glutamate receptors. Activation of NMDA receptors results in the opening of the ion channel that is nonselective to cations, with a combined reversal potential near 0 mV. While the opening and closing of the ion channel is primarily gated by ligand binding, the current flow through the ion channel is voltage-dependent. Specifically located on the receptor, extracellular magnesium (Mg2+) and zinc (Zn2+) ions can bind and prevent other cations from flowing through the open ion channel. A voltage-dependent flow of predominantly calcium (Ca2+), sodium (Na+), and potassium (K+) ions into and out of the cell is made possible by the depolarization of the cell, which displaces and repels the Mg2+ and Zn2+ ions from the pore.[8][9][10][11] Ca2+ flux through NMDA receptors in particular is thought to be critical in synaptic plasticity, a cellular mechanism for learning and memory, due to proteins which bind to and are activated by Ca2+ ions.
Activity of the NMDA receptor is blocked by many psychoactive drugs such as phencyclidine (PCP), alcohol (ethanol) and dextromethorphan (DXM). The anaesthetic and analgesic effects of the drugs ketamine and nitrous oxide are also partially due to their effects at blocking NMDA receptor activity. In contrast, overactivation of NMDAR by NMDA agonists increases the cytosolic concentrations of calcium and zinc, which significantly contributes to neural death, an effect known to be prevented by cannabinoids, mediated by activation of the CB1 receptor, which leads HINT1 protein to counteract the toxic effects of NMDAR-mediated NO production and zinc release.[12] As well as preventing methamphetamine-induced neurotoxicity via inhibition of nitric oxide synthase (nNOS) expression and astrocyte activation, it is seen to reduce methamphetamine induced brain damage through CB1-dependent and independent mechanisms, respectively, and inhibition of methamphetamine induced astrogliosis is likely to occur through a CB2 receptor dependent mechanism for THC.[13] Since 1989, memantine has been recognized to be an uncompetitive antagonist of the NMDA receptor, entering the channel of the receptor after it has been activated and thereby blocking the flow of ions.[14][15][16]
Overactivation of the receptor, causing excessive influx of Ca2+ can lead to excitotoxicity which is implied to be involved in some neurodegenerative disorders. Blocking of NMDA receptors could therefore, in theory, be useful in treating such diseases.[16][17][18][19] However, hypofunction of NMDA receptors (due to glutathione deficiency or other causes) may be involved in impairment of synaptic plasticity[20] and could have other negative repercussions. The main problem with the utilization of NMDA receptor antagonists for neuroprotection is that the physiological actions of the NMDA receptor are essential for normal neuronal function. To be clinically useful NMDA antagonists need to block excessive activation without interfering with normal functions. Memantine has this property.[21]
- ^ Laube B, Hirai H, Sturgess M, Betz H, Kuhse J (March 1997). "Molecular determinants of agonist discrimination by NMDA receptor subunits: analysis of the glutamate binding site on the NR2B subunit". Neuron. 18 (3): 493–503. doi:10.1016/S0896-6273(00)81249-0. PMID 9115742.
Since two molecules of glutamate and glycine each are thought to be required for channel activation (3, 6), this implies that the NMDA receptor should be composed of at least four subunits.
- ^ Anson LC, Chen PE, Wyllie DJ, Colquhoun D, Schoepfer R (January 1998). "Identification of amino acid residues of the NR2A subunit that control glutamate potency in recombinant NR1/NR2A NMDA receptors". The Journal of Neuroscience. 18 (2): 581–589. doi:10.1523/JNEUROSCI.18-02-00581.1998. PMC 6792534. PMID 9425000.
- ^ Vyklicky, V.; Korinek, M.; Smejkalova, T.; Balik, A.; Krausova, B.; Kaniakova, M.; Lichnerova, K.; Cerny, J.; Krusek, J.; Dittert, I.; Horak, M.; Vyklicky, L. (2014). "Structure, function, and pharmacology of NMDA receptor channels". Physiological Research. 63 (Suppl 1): S191–203. doi:10.33549/physiolres.932678. ISSN 1802-9973. PMID 24564659.
- ^ Jewett, Benjamin E.; Thapa, Bicky (2024), "Physiology, NMDA Receptor", StatPearls, Treasure Island (FL): StatPearls Publishing, PMID 30137779, retrieved 2024-03-04
- ^ Cite error: The named reference
Furukawawas invoked but never defined (see the help page). - ^ Li F, Tsien JZ (July 2009). "Memory and the NMDA receptors". The New England Journal of Medicine. 361 (3): 302–303. doi:10.1056/NEJMcibr0902052. PMC 3703758. PMID 19605837.
- ^ Moriyoshi K, Masu M, Ishii T, Shigemoto R, Mizuno N, Nakanishi S (November 1991). "Molecular cloning and characterization of the rat NMDA receptor". Nature. 354 (6348): 31–37. Bibcode:1991Natur.354...31M. doi:10.1038/354031a0. PMID 1834949. S2CID 4368947.
- ^ Dingledine R, Borges K, Bowie D, Traynelis SF (March 1999). "The glutamate receptor ion channels". Pharmacological Reviews. 51 (1): 7–61. PMID 10049997.
- ^ Liu Y, Zhang J (October 2000). "Recent development in NMDA receptors". Chinese Medical Journal. 113 (10): 948–956. PMID 11775847.
- ^ Cull-Candy S, Brickley S, Farrant M (June 2001). "NMDA receptor subunits: diversity, development and disease". Current Opinion in Neurobiology. 11 (3): 327–335. doi:10.1016/S0959-4388(00)00215-4. PMID 11399431. S2CID 11929361.
- ^ Paoletti P, Neyton J (February 2007). "NMDA receptor subunits: function and pharmacology" (PDF). Current Opinion in Pharmacology. 7 (1): 39–47. doi:10.1016/j.coph.2006.08.011. PMID 17088105.
- ^ Sánchez-Blázquez P, Rodríguez-Muñoz M, Vicente-Sánchez A, Garzón J (November 2013). "Cannabinoid receptors couple to NMDA receptors to reduce the production of NO and the mobilization of zinc induced by glutamate". Antioxidants & Redox Signaling. 19 (15): 1766–1782. doi:10.1089/ars.2012.5100. PMC 3837442. PMID 23600761.
- ^ Castelli MP, Madeddu C, Casti A, Casu A, Casti P, Scherma M, et al. (2014-05-20). "Δ9-tetrahydrocannabinol prevents methamphetamine-induced neurotoxicity". PLOS ONE. 9 (5): e98079. Bibcode:2014PLoSO...998079C. doi:10.1371/journal.pone.0098079. PMC 4028295. PMID 24844285.
- ^ Johnson JW, Kotermanski SE (February 2006). "Mechanism of action of memantine". Current Opinion in Pharmacology. 6 (1): 61–67. doi:10.1016/j.coph.2005.09.007. PMID 16368266.
- ^ Dominguez E, Chin TY, Chen CP, Wu TY (December 2011). "Management of moderate to severe Alzheimer's disease: focus on memantine". Taiwanese Journal of Obstetrics & Gynecology. 50 (4): 415–423. doi:10.1016/j.tjog.2011.10.004. PMID 22212311.
- ^ a b Chen HS, Lipton SA (June 2006). "The chemical biology of clinically tolerated NMDA receptor antagonists". Journal of Neurochemistry. 97 (6): 1611–1626. doi:10.1111/j.1471-4159.2006.03991.x. PMID 16805772. S2CID 18376541.
- ^ Kemp JA, McKernan RM (November 2002). "NMDA receptor pathways as drug targets". Nature Neuroscience. 5 (11): 1039–1042. doi:10.1038/nn936. PMID 12403981. S2CID 41383776.
- ^ Lipton SA (February 2006). "Paradigm shift in neuroprotection by NMDA receptor blockade: memantine and beyond". Nature Reviews. Drug Discovery. 5 (2): 160–170. doi:10.1038/nrd1958. PMID 16424917. S2CID 21379258.
- ^ Koch HJ, Szecsey A, Haen E (1 January 2004). "NMDA-antagonism (memantine): an alternative pharmacological therapeutic principle in Alzheimer's and vascular dementia". Current Pharmaceutical Design. 10 (3): 253–259. doi:10.2174/1381612043386392. PMID 14754385.
- ^ Steullet P, Neijt HC, Cuénod M, Do KQ (February 2006). "Synaptic plasticity impairment and hypofunction of NMDA receptors induced by glutathione deficit: relevance to schizophrenia". Neuroscience. 137 (3): 807–819. doi:10.1016/j.neuroscience.2005.10.014. PMID 16330153. S2CID 1417873.
- ^ Lipton SA (January 2004). "Failures and successes of NMDA receptor antagonists: molecular basis for the use of open-channel blockers like memantine in the treatment of acute and chronic neurologic insults". NeuroRx. 1 (1): 101–110. doi:10.1602/neurorx.1.1.101. PMC 534915. PMID 15717010.