 | |
| Clinical data | |
|---|---|
| Pronunciation | ne TAR soo dil |
| Trade names | Rhopressa, Rhokiinsa |
| Other names | AR-13324 |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a618014 |
| License data |
|
| Routes of administration | Eye drops, topical |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Metabolism | Esterases in the cornea |
| Metabolites | AR-13503 (active metabolite) |
| Elimination half-life | 16–17 hrs |
| Duration of action | ≥ 24 hrs |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.251.524 |
| Chemical and physical data | |
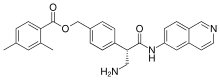
| Formula | C28H27N3O3 |
| Molar mass | 453.542 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Netarsudil, sold under the brand name Rhopressa among others, is a medication for the treatment of glaucoma.[1][2][3] In the United States, in December 2017, the Food and Drug Administration (FDA) approved a 0.02% ophthalmic solution for the lowering of elevated intraocular pressure in people with open-angle glaucoma or ocular hypertension.[4][5] The European Medicines Agency approved it in 2019 for the same uses under the brand name Rhokiinsa.[2]
The FDA considers it to be a first-in-class medication.[6]
- ^ a b "Rhopressa- netarsudil solution/ drops". DailyMed. Retrieved 2 May 2021.
- ^ a b c "Rhokiinsa EPAR". European Medicines Agency (EMA). 16 September 2019. Retrieved 27 September 2020.
- ^ Dasso L, Al-Khaled T, Sonty S, Aref AA (2018). "Profile of netarsudil ophthalmic solution and its potential in the treatment of open-angle glaucoma: evidence to date". Clinical Ophthalmology. 12. Auckland, N.Z.: 1939–1944. doi:10.2147/OPTH.S154001. PMC 6177382. PMID 30323550.
- ^ "Rhopressa (netarsudil) Ophthalmic Solution". U.S. Food and Drug Administration (FDA). 29 January 2018. Retrieved 3 June 2020.
- ^ "Aerie (AERI) Gets Early FDA Approval for Lead Drug Rhopressa". 19 December 2017.
- ^ New Drug Therapy Approvals 2017 (PDF). U.S. Food and Drug Administration (FDA) (Report). January 2018. Retrieved 16 September 2020.