 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Niclocide, Fenasal, Phenasal, others[1] |
| AHFS/Drugs.com | Micromedex Detailed Consumer Information |
| Routes of administration | By mouth |
| ATC code | |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.000.052 |
| Chemical and physical data | |
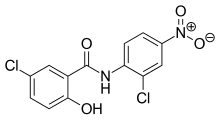
| Formula | C13H8Cl2N2O4 |
| Molar mass | 327.12 g·mol−1 |
| 3D model (JSmol) | |
| Melting point | 225 to 230 °C (437 to 446 °F) |
| |
| |
| | |
Niclosamide, sold under the brand name Niclocide among others, is an anthelmintic medication used to treat tapeworm infestations, including diphyllobothriasis, hymenolepiasis, and taeniasis. It is not effective against other worms such as flukes or roundworms.[2] It is taken by mouth.[3]
Side effects include nausea, vomiting, abdominal pain, and itchiness. It may be used during pregnancy.[3] It works by blocking glucose uptake and oxidative phosphorylation by the worm.[4]
Niclosamide was first synthesized in 1958.[5] It is on the World Health Organization's List of Essential Medicines.[6] Niclosamide is not available for human use in the United States.[7]
- ^ CID 4477 from PubChem
- ^ "Niclosamide Advanced Patient Information - Drugs.com". www.drugs.com. Archived from the original on 20 December 2016. Retrieved 8 December 2016.
- ^ a b World Health Organization (2009). Stuart MC, Kouimtzi M, Hill SR (eds.). WHO Model Formulary 2008. World Health Organization. pp. 81, 87, 591. hdl:10665/44053. ISBN 9789241547659.
- ^ Lanusse CE, Alvarez LI, Sallovitz JM, Mottier ML, Sanchez Bruni SF (13 May 2013). "Antinematodal Drugs". In Riviere JE, Papich MG (eds.). Veterinary Pharmacology and Therapeutics. John Wiley & Sons. p. 1096. ISBN 978-1-118-68590-7. Archived from the original on 10 September 2017.
- ^ Mehlhorn H (2008). Encyclopedia of Parasitology: A-M. Springer Science & Business Media. p. 483. ISBN 978-3-540-48994-8. Archived from the original on 2016-12-20.
- ^ World Health Organization (2019). World Health Organization model list of essential medicines: 21st list 2019. Geneva: World Health Organization. hdl:10665/325771. WHO/MVP/EMP/IAU/2019.06. License: CC BY-NC-SA 3.0 IGO.
- ^ "Dipylidium – Resources for Health Professionals". U.S. Centers for Disease Control and Prevention (CDC). 21 May 2020.