 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Zyprexa, others[1] |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a601213 |
| License data |
|
| Pregnancy category |
|
| Routes of administration | By mouth, intramuscular injection |
| Drug class | Atypical antipsychotic |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | 60–65%[8][9][10] |
| Protein binding | 93%[11] |
| Metabolism | Liver (direct glucuronidation and CYP1A2 mediated oxidation) |
| Elimination half-life | 33 hours, 51.8 hours (elderly)[11] |
| Excretion | Urine (57%; 7% as unchanged drug), faeces (30%)[11][12] |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.125.320 |
| Chemical and physical data | |
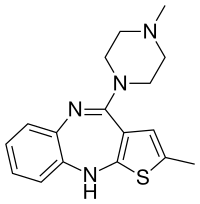
| Formula | C17H20N4S |
| Molar mass | 312.44 g·mol−1 |
| 3D model (JSmol) | |
| Melting point | 195 °C (383 °F) |
| Solubility in water | Practically insoluble in water |
| |
| |
| | |
Olanzapine, sold under the brand name Zyprexa among others, is an atypical antipsychotic primarily used to treat schizophrenia and bipolar disorder.[13] It is also sometimes used off-label for treatment of chemotherapy-induced nausea and vomiting[14] and as an appetite stimulant.[15] For schizophrenia, it can be used for both new-onset disease and long-term maintenance.[13] It is taken by mouth or by injection into a muscle.[13]
Common side effects include significant weight gain, feeling tired, dizziness, constipation, dry mouth, and restlessness.[13] Other side effects include low blood pressure with standing, allergic reactions, neuroleptic malignant syndrome, high blood sugar, seizures, and tardive dyskinesia.[13] In older people with dementia, its use increases the risk of death.[13] Use in the later part of pregnancy may result in a movement disorder in the baby for some time after birth.[13] Although how it works is not entirely clear, it blocks dopamine and serotonin receptors.[13]
Olanzapine was patented in 1991 and approved for medical use in the United States in 1996.[13][16] It is available as a generic medication.[13] In 2022, it was the 171st most commonly prescribed medication in the United States, with more than 3 million prescriptions.[17][18] It is on the World Health Organization's List of Essential Medicines.[19]
Eli Lilly also markets olanzapine in a fixed-dose combination with fluoxetine as olanzapine/fluoxetine (Symbyax), which was approved by the US FDA for the treatment of depressive episodes of bipolar I disorder in 2003 and for treatment-resistant depression in 2009.[20]
Alkermes also markets olanzapine in a fixed-dose combination with samidorphan as olanzapine/samidorphan (Lybalvi), which was approved by the US FDA for the treatment of schizophrenia and bipolar I disorder in May 2021. Lybalvi suppresses the metabolic side effect of olanzapine by nearly 2%.[21]
- ^ Cite error: The named reference
Drugnameswas invoked but never defined (see the help page). - ^ "FDA-sourced list of all drugs with black box warnings (Use Download Full Results and View Query links.)". nctr-crs.fda.gov. FDA. Retrieved 22 October 2023.
- ^ Anvisa (31 March 2023). "RDC Nº 784 – Listas de Substâncias Entorpecentes, Psicotrópicas, Precursoras e Outras sob Controle Especial" [Collegiate Board Resolution No. 784 – Lists of Narcotic, Psychotropic, Precursor, and Other Substances under Special Control] (in Brazilian Portuguese). Diário Oficial da União (published 4 April 2023). Archived from the original on 3 August 2023. Retrieved 16 August 2023.
- ^ Cite error: The named reference
EMCwas invoked but never defined (see the help page). - ^ "Zyprexa- olanzapine tablet; Zyprexa Zydis- olanzapine tablet, orally disintegrating; Zyprexa intramuscular- olanzapine injection, powder, for solution". DailyMed. 13 December 2023. Retrieved 17 October 2024.
- ^ "Zyprexa EPAR". European Medicines Agency (EMA). 27 September 1996. Retrieved 27 February 2024.
- ^ "Zypadhera". European Medicines Agency (EMA). 19 November 2008. Retrieved 26 October 2024.
- ^ Kassahun K, Mattiuz E, Nyhart E, Obermeyer B, Gillespie T, Murphy A, et al. (January 1997). "Disposition and biotransformation of the antipsychotic agent olanzapine in humans". Drug Metabolism and Disposition. 25 (1): 81–93. PMID 9010634.
- ^ Callaghan JT, Bergstrom RF, Ptak LR, Beasley CM (September 1999). "Olanzapine. Pharmacokinetic and pharmacodynamic profile". Clinical Pharmacokinetics. 37 (3): 177–193. doi:10.2165/00003088-199937030-00001. PMID 10511917.
- ^ Mauri MC, Volonteri LS, Colasanti A, Fiorentini A, De Gaspari IF, Bareggi SR (2007). "Clinical pharmacokinetics of atypical antipsychotics: a critical review of the relationship between plasma concentrations and clinical response". Clinical Pharmacokinetics. 46 (5): 359–388. doi:10.2165/00003088-200746050-00001. PMID 17465637. S2CID 43859718.
- ^ a b c Cite error: The named reference
TGAwas invoked but never defined (see the help page). - ^ Cite error: The named reference
MSRwas invoked but never defined (see the help page). - ^ a b c d e f g h i j "Olanzapine, Olanzapine Pamoate Monograph for Professionals". Drugs.com. AHFS. Retrieved 24 December 2018.
- ^ Razvi Y, Chan S, McFarlane T, McKenzie E, Zaki P, DeAngelis C, et al. (January 2019). "ASCO, NCCN, MASCC/ESMO: a comparison of antiemetic guidelines for the treatment of chemotherapy-induced nausea and vomiting in adult patients". Supportive Care in Cancer. 27 (1): 87–95. doi:10.1007/s00520-018-4464-y. PMID 30284039.
- ^ Sandhya L, Devi Sreenivasan N, Goenka L, Dubashi B, Kayal S, Solaiappan M, et al. (May 2023). "Randomized Double-Blind Placebo-Controlled Study of Olanzapine for Chemotherapy-Related Anorexia in Patients With Locally Advanced or Metastatic Gastric, Hepatopancreaticobiliary, and Lung Cancer". Journal of Clinical Oncology. 41 (14): 2617–2627. doi:10.1200/JCO.22.01997. PMID 36977285.
- ^ Taylor D, Paton C, Kapur S (2015). The Maudsley Prescribing Guidelines in Psychiatry (12th ed.). London, U K: Wiley-Blackwell. p. 16. ISBN 978-1-118-75460-3.
- ^ "The Top 300 of 2022". ClinCalc. Archived from the original on 30 August 2024. Retrieved 30 August 2024.
- ^ "Olanzapine Drug Usage Statistics, United States, 2013 - 2022". ClinCalc. Retrieved 30 August 2024.
- ^ World Health Organization (2023). The selection and use of essential medicines 2023: web annex A: World Health Organization model list of essential medicines: 23rd list (2023). Geneva: World Health Organization. hdl:10665/371090. WHO/MHP/HPS/EML/2023.02.
- ^ "Symbyax- olanzapine and fluoxetine hydrochloride capsule". DailyMed. 21 April 2020. Retrieved 30 September 2020.
- ^ "DailyMed - Lybalvi- olanzapine and samidorphan l-malate tablet, film coated". DailyMed.