 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Caverject, Muse, others |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a695022 |
| License data |
|
| Routes of administration | Intravenous |
| ATC code | |
| Legal status | |
| Legal status | |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.010.925 |
| Chemical and physical data | |
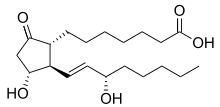
| Formula | C20H34O5 |
| Molar mass | 354.487 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
Prostaglandin E1 (PGE1) is a naturally occurring prostaglandin and is also used as a medication (alprostadil).[2]
In infants with congenital heart defects, it is delivered by slow injection into a vein to open the ductus arteriosus until surgery can be carried out.[3] By injection into the penis or placement in the urethra, it is used to treat erectile dysfunction.[4]
Common side effects when given to babies include decreased breathing, fever, and low blood pressure.[2] When injected into the penis for erectile dysfunction; side effects may include penile pain, bleeding at the site of injection, and prolonged erection (priapism).[2] Prostaglandin E1 is in the vasodilator family of medications.[2] It works by opening blood vessels and relaxing smooth muscle.[2]
Prostaglandin E1 was isolated in 1957 and approved for medical use in the United States in 1981.[2][5] It is on the World Health Organization's List of Essential Medicines.[6]
- ^ "FDA-sourced list of all drugs with black box warnings (Use Download Full Results and View Query links.)". nctr-crs.fda.gov. FDA. Retrieved 22 October 2023.
- ^ a b c d e f "Alprostadil". The American Society of Health-System Pharmacists. Archived from the original on 16 January 2017. Retrieved 8 January 2017.
- ^ Northern Neonatal Network (208). Neonatal Formulary: Drug Use in Pregnancy and the First Year of Life (5 ed.). John Wiley & Sons. p. 2010. ISBN 9780470750353. Archived from the original on 13 January 2017.
- ^ British National Formulary (BNF) (69th ed.). British Medical Association. 2015. p. 569. ISBN 9780857111562.
- ^ Sneader W (2005). Drug Discovery: A History. John Wiley & Sons. p. 185. ISBN 9780470015520. Archived from the original on 13 January 2017.
- ^ World Health Organization (2019). World Health Organization model list of essential medicines: 21st list 2019. Geneva: World Health Organization. hdl:10665/325771. WHO/MVP/EMP/IAU/2019.06. License: CC BY-NC-SA 3.0 IGO.