 | |
| Clinical data | |
|---|---|
| Trade names | Cisobitan |
| Other names | Quadrosilane; KABI-1774; 2,6-cisdiphenylhexa- methylcyclotetrasiloxane |
| Drug class | Nonsteroidal estrogen |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
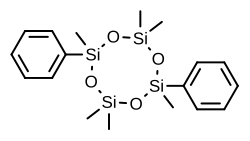
| Formula | C18H28O4Si4 |
| Molar mass | 420.758 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Quadrosilan (INN, BAN) (brand name Cisobitan; former developmental code name KABI-1774) is a synthetic nonsteroidal estrogen that was developed in the 1970s and that is or has been used as an antigonadotropic agent in the treatment of prostate cancer.[1][2][3][4] It is an organosilicon compound, and is also known as 2,6-cisdiphenylhexamethylcyclotetrasiloxane.[3][5] Quadrosilan has estrogenic activity equivalent to that of estradiol,[6] and can produce feminization and gynecomastia as side effects in male patients.[7][8]
- ^ Elks J (14 November 2014). The Dictionary of Drugs: Chemical Data: Chemical Data, Structures and Bibliographies. Springer. pp. 629–. ISBN 978-1-4757-2085-3.
- ^ Morton IK, Hall JM (6 December 2012). Concise Dictionary of Pharmacological Agents: Properties and Synonyms. Springer Science & Business Media. pp. 243–. ISBN 978-94-011-4439-1.
- ^ a b Alfthan O, Andersson L, Esposti PL, Fosså SD, Gammelgaard PA, Gjöres JE, et al. (1983). "Cisobitan in treatment of prostatic cancer. A prospective controlled multicentre study". Scandinavian Journal of Urology and Nephrology. 17 (1): 37–43. doi:10.3109/00365598309179778. PMID 6346476.
- ^ Chisholm GD (1985). "Treatment of advanced cancer of the prostate". Seminars in Surgical Oncology. 1 (1): 38–55. doi:10.1002/ssu.2980010106. PMID 3887539.
- ^ Apeloig Y (1989). The Chemistry of Organic Silicon Compounds. John Wiley & Sons Canada, Limited. p. 1154. ISBN 978-0-471-91993-3.
- ^ Mills JS, Showell GA (September 2004). "Exploitation of silicon medicinal chemistry in drug discovery". Expert Opinion on Investigational Drugs. 13 (9): 1149–1157. doi:10.1517/13543784.13.9.1149. PMID 15330746. S2CID 26669175.
- ^ Strindberg B (1978). "Biochemical Effects of 2, 6-cis-Diphenylhexamethylcyclotetrasiloxane in Man". Biochemistry of Silicon and Related Problems. pp. 515–520. doi:10.1007/978-1-4613-4018-8_23. ISBN 978-1-4613-4020-1.
- ^ Krarup T, Rasmussen F, Gammelgaard PA (1978). "Prostatic carcinoma treated with 2,6-cis-diphenylhexamethylcyclotetrasiloxane (Cisobitan)". Scandinavian Journal of Urology and Nephrology. 12 (1): 11–15. PMID 345431.