 | |
| Clinical data | |
|---|---|
| Other names | ALKS-33; RDC-0313; 3-Carboxamido-4-hydroxynaltrexone |
| Routes of administration | Oral |
| Pharmacokinetic data | |
| Elimination half-life | 7–9 hours[1][2] |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| KEGG | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
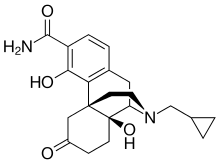
| Formula | C21H26N2O4 |
| Molar mass | 370.449 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Samidorphan (INN, USAN) (developmental code names ALKS-33, RDC-0313) is an opioid antagonist that in the form of olanzapine/samidorphan (brand name Lybalvi) is used in the treatment of schizophrenia and bipolar disorder.[1][3][4] Samidorphan reduces the weight gain associated with olanzapine.[5][6] Samidorphan is taken by mouth.[1][3]
Samidorphan was under development as a standalone medication for various indications but has been discontinued.[7] Buprenorphine/samidorphan for the treatment of major depressive disorder was rejected by the Food and Drug Administration due to insufficient evidence of effectiveness but remains in preregistration as of September 2021.[8] Development of baclofen/samidorphan has also been discontinued.[9]
- ^ a b c Chaudhary AM, Khan MF, Dhillon SS, Naveed S (July 2019). "A Review of Samidorphan: A Novel Opioid Antagonist". Cureus. 11 (7): e5139. doi:10.7759/cureus.5139. PMC 6741386. PMID 31523568.
- ^ Turncliff R, DiPetrillo L, Silverman B, Ehrich E (February 2015). "Single- and multiple-dose pharmacokinetics of samidorphan, a novel opioid antagonist, in healthy volunteers". Clinical Therapeutics. 37 (2): 338–348. doi:10.1016/j.clinthera.2014.10.001. PMID 25456560.
- ^ a b "LYBALVI: Highlight of Prescribing Information" (PDF). U.S. Food and Drug Administration.
- ^ "Olanzapine/samidorphan - Alkermes plc". Adis Insight. Springer Nature Switzerland AG.
- ^ Citrome L, Graham C, Simmons A, Jiang Y, Todtenkopf MS, Silverman B, et al. (2021). "An Evidence-Based Review of OLZ/SAM for Treatment of Adults with Schizophrenia or Bipolar I Disorder". Neuropsychiatric Disease and Treatment. 17: 2885–2904. doi:10.2147/NDT.S313840. PMC 8437420. PMID 34526769.
- ^ Paik J (August 2021). "Olanzapine/Samidorphan: First Approval". Drugs. 81 (12): 1431–1436. doi:10.1007/s40265-021-01568-0. PMID 34304374. S2CID 236215923.
- ^ "Samidorphan". Adis Insight. Springer Nature Switzerland AG.
- ^ "Buprenorphine/samidorphan - Alkermes". Adis Insight. Springer Nature Switzerland AG.
- ^ "Baclofen/samidorphan". Adis Insight. Springer Nature Switzerland AG.