| |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name
3,4-Dihydroxycyclobut-3-ene-1,2-dione | |||
| Other names
Quadratic acid
Cyclobutenedioic acid | |||
| Identifiers | |||
3D model (JSmol)
|
|||
| ChemSpider | |||
| ECHA InfoCard | 100.018.875 | ||
| EC Number |
| ||
PubChem CID
|
|||
| UNII | |||
CompTox Dashboard (EPA)
|
|||
| |||
| |||
| Properties | |||
| C4H2O4 | |||
| Molar mass | 114.056 g·mol−1 | ||
| Appearance | white crystalline powder | ||
| Melting point | > 300 °C (572 °F; 573 K) | ||
| Acidity (pKa) | pKa1 = 1.5 pKa2 = 3.4 | ||
| Hazards[2] | |||
| GHS labelling: | |||

| |||
| Danger | |||
| H314 | |||
| P260, P280, P301+P330+P331, P303+P361+P353, P304+P340+P310, P305+P351+P338 | |||
| Flash point | 190 °C (374 °F; 463 K)[3] | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
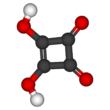
Squaric acid, also called quadratic acid because its four carbon atoms approximately form a square, is a diprotic organic acid with the chemical formula C4O2(OH)2.[4]
The conjugate base of squaric acid is the hydrogensquarate anion HC4O−4; and the conjugate base of the hydrogensquarate anion is the divalent squarate anion C4O2−4. This is one of the oxocarbon anions, which consist only of carbon and oxygen.
Squaric acid is a reagent for chemical synthesis, used for instance to make photosensitive squaraine dyes and inhibitors of protein tyrosine phosphatases.
- ^ 3,4-Dihydroxy-3-cyclobutene-1,2-dione. Sigma-Aldrich
- ^ "SICHERHEITSDATENBLATT". 21 March 2021.
- ^ 3,4-Dihydroxy-3-cyclobutene-1,2-dione, 98+%. Alfa Aesar
- ^ Robert West (1980). "History of the Oxocarbons". In Robert West (ed.). Oxocarbons. Academic Press. pp. 1–14. doi:10.1016/B978-0-12-744580-9.50005-1. ISBN 9780127445809.