 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Belsomra |
| Other names | MK-4305; MK4305 |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a614046 |
| License data |
|
| Pregnancy category |
|
| Dependence liability | Low |
| Addiction liability | Low |
| Routes of administration | By mouth[2] |
| Drug class | Orexin receptor antagonist; Hypnotic; Sedative |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | 82% (10 mg; lower at higher doses)[2][5] |
| Protein binding | 99.5%[6][2] |
| Metabolism | Liver (CYP3A major, CYP2C19 minor)[2] |
| Metabolites | Hydroxysuvorexant (inactive)[2] |
| Elimination half-life | 12.2 hours (8–19 hours) (40 mg)[2][5][7] |
| Excretion | Feces: 66%[2] Urine: 23%[2] |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.210.546 |
| Chemical and physical data | |
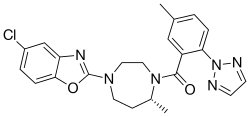
| Formula | C23H23ClN6O2 |
| Molar mass | 450.93 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| (verify) | |
Suvorexant, sold under the brand name Belsomra, is an orexin antagonist medication which is used in the treatment of insomnia.[2][6] It is indicated specifically for the treatment of insomnia characterized by difficulties with sleep onset and/or maintenance in adults.[2][6] Suvorexant helps with falling asleep faster, sleeping longer, being awake less in the middle of the night, and having better quality of sleep.[2][8] Its effectiveness is modest,[9] and is similar to that of other orexin antagonists, but is lower than that of benzodiazepines and Z-drugs.[10] Suvorexant is taken by mouth.[2][11][6]
Side effects of suvorexant include somnolence, daytime sleepiness and sedation, headache, dizziness, abnormal dreams, dry mouth, and impaired next-day driving ability.[2][8][12] Rarely, sleep paralysis, sleep-related hallucinations, complex sleep behaviors like sleepwalking, and suicidal ideation may occur.[2][6][9] Tolerance, dependence, withdrawal, and rebound effects do not appear to occur significantly with the medication.[2][13][14] Suvorexant is a dual orexin receptor antagonist (DORA).[6] It acts as a selective dual antagonist of the orexin OX1 and OX2 receptors.[6] The medication has an intermediate elimination half-life of 12 hours and a time to peak of about 2 to 3 hours.[2][6] Unlike benzodiazepines and Z-drugs, suvorexant does not interact with GABA receptors, instead having a distinct mechanism of action.[6][15]
Clinical development of suvorexant began in 2006[16] and it was introduced for medical use in 2014.[2][17] The medication is a schedule IV controlled substance in the United States and may have a modest potential for misuse.[18][2][19] In other places, such as Australia, suvorexant is a prescription-only medicine and is not a controlled drug.[1] Suvorexant is not available in generic formulations.[11][20][21] Besides suvorexant, other orexin receptor antagonists like lemborexant and daridorexant have also been introduced.[22][23]
- ^ a b Cite error: The named reference
Belsomra-AU-Labelwas invoked but never defined (see the help page). - ^ a b c d e f g h i j k l m n o p q r s "Belsomra- suvorexant tablet, film coated". DailyMed. Retrieved 30 January 2020.
- ^ "Prescription medicines: registration of new chemical entities in Australia, 2016". Therapeutic Goods Administration (TGA). 21 June 2022. Retrieved 10 April 2023.
- ^ "Regulatory Decision Summary for Belsomra". 23 October 2014.
- ^ a b Cite error: The named reference
MerckSharpDohme2013was invoked but never defined (see the help page). - ^ a b c d e f g h i Jacobson LH, Callander GE, Hoyer D (November 2014). "Suvorexant for the treatment of insomnia". Expert Review of Clinical Pharmacology. 7 (6): 711–730. doi:10.1586/17512433.2014.966813. PMID 25318834. S2CID 41099462.
- ^ Cite error: The named reference
DimovaBrarMen2014was invoked but never defined (see the help page). - ^ a b Cite error: The named reference
pmid28365447was invoked but never defined (see the help page). - ^ a b Cite error: The named reference
pmid26648692was invoked but never defined (see the help page). - ^ Cite error: The named reference
pmid35843245was invoked but never defined (see the help page). - ^ a b Cite error: The named reference
Drugs.com-Belsomra-Genericwas invoked but never defined (see the help page). - ^ Kishi T, Matsunaga S, Iwata N (2015). "Suvorexant for Primary Insomnia: A Systematic Review and Meta-Analysis of Randomized Placebo-Controlled Trials". PLOS ONE. 10 (8): e0136910. Bibcode:2015PLoSO..1036910K. doi:10.1371/journal.pone.0136910. PMC 4552781. PMID 26317363.
- ^ Cite error: The named reference
pmid28994603was invoked but never defined (see the help page). - ^ Cite error: The named reference
pmid32901578was invoked but never defined (see the help page). - ^ Atkin T, Comai S, Gobbi G (April 2018). "Drugs for Insomnia beyond Benzodiazepines: Pharmacology, Clinical Applications, and Discovery". Pharmacological Reviews. 70 (2): 197–245. doi:10.1124/pr.117.014381. PMID 29487083. S2CID 3578916.
- ^ Cite error: The named reference
pmid25406050was invoked but never defined (see the help page). - ^ Cite error: The named reference
APDNews2014was invoked but never defined (see the help page). - ^ Cite error: The named reference
FederalRegister2016was invoked but never defined (see the help page). - ^ Cite error: The named reference
pmid25231363was invoked but never defined (see the help page). - ^ Cite error: The named reference
pmid30092886was invoked but never defined (see the help page). - ^ Cite error: The named reference
pmid26955275was invoked but never defined (see the help page). - ^ Cite error: The named reference
pmid35043499was invoked but never defined (see the help page). - ^ Cite error: The named reference
pmid35298826was invoked but never defined (see the help page).