| |||

| |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name
2-Methylpropan-2-ol | |||
Other names
| |||
| Identifiers | |||
3D model (JSmol)
|
|||
| 906698 | |||
| ChEBI | |||
| ChEMBL | |||
| ChemSpider | |||
| DrugBank | |||
| ECHA InfoCard | 100.000.809 | ||
| EC Number |
| ||
| 1833 | |||
| MeSH | tert-Butyl+Alcohol | ||
PubChem CID
|
|||
| RTECS number |
| ||
| UNII | |||
| UN number | 1120 | ||
CompTox Dashboard (EPA)
|
|||
| |||
| |||
| Properties | |||
| C4H10O | |||
| Molar mass | 74.123 g·mol−1 | ||
| Appearance | Colorless solid | ||
| Odor | Camphorous | ||
| Density | 0.775 g/mL | ||
| Melting point | 25 to 26 °C; 77 to 79 °F; 298 to 299 K | ||
| Boiling point | 82 to 83 °C; 179 to 181 °F; 355 to 356 K | ||
| miscible[2] | |||
| log P | 0.584 | ||
| Vapor pressure | 4.1 kPa (at 20 °C) | ||
| Acidity (pKa) | 16.54 [3] | ||
| 5.742×10−5 cm3/mol | |||
Refractive index (nD)
|
1.387 | ||
| 1.31 D | |||
| Thermochemistry | |||
Heat capacity (C)
|
215.37 J K−1 mol−1 | ||
Std molar
entropy (S⦵298) |
189.5 J K−1 mol−1 | ||
Std enthalpy of
formation (ΔfH⦵298) |
−360.04 to −358.36 kJ mol−1 | ||
Std enthalpy of
combustion (ΔcH⦵298) |
−2.64479 to −2.64321 MJ mol−1 | ||
| Hazards | |||
| GHS labelling: | |||
 
| |||
| Danger | |||
| H225, H319, H332, H335 | |||
| P210, P261, P305+P351+P338 | |||
| NFPA 704 (fire diamond) | |||
| Flash point | 11 °C (52 °F; 284 K) | ||
| 480 °C (896 °F; 753 K) | |||
| Explosive limits | 2.4–8.0% | ||
| Lethal dose or concentration (LD, LC): | |||
LD50 (median dose)
|
3559 mg/kg (rabbit, oral) 3500 mg/kg (rat, oral)[4] | ||
| NIOSH (US health exposure limits): | |||
PEL (Permissible)
|
TWA 100 ppm (300 mg/m3)[1] | ||
REL (Recommended)
|
TWA 100 ppm (300 mg/m3) ST 150 ppm (450 mg/m3)[1] | ||
IDLH (Immediate danger)
|
1600 ppm[1] | ||
| Safety data sheet (SDS) | inchem.org | ||
| Related compounds | |||
Related butanols
|
2-Butanol | ||
Related compounds
|
2-Methyl-2-butanol Trimethylsilanol | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
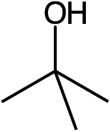
tert-Butyl alcohol is the simplest tertiary alcohol, with a formula of (CH3)3COH (sometimes represented as t-BuOH). Its isomers are 1-butanol, isobutanol, and butan-2-ol. tert-Butyl alcohol is a colorless solid, which melts near room temperature and has a camphor-like odor. It is miscible with water, ethanol and diethyl ether.
- ^ a b c d NIOSH Pocket Guide to Chemical Hazards. "#0078". National Institute for Occupational Safety and Health (NIOSH).
- ^ "ICSC 0114 – tert-Butanol". Inchem.org. Retrieved 29 March 2018.
- ^ Reeve, W.; Erikson, C. M.; Aluotto, P. F. (1979). "tert-Butyl alcohol". Can. J. Chem. 57: 2747. doi:10.1139/v79-444.
- ^ "tert-Butyl alcohol". Immediately Dangerous to Life or Health Concentrations (IDLH). National Institute for Occupational Safety and Health (NIOSH).