 | |
 | |
| Clinical data | |
|---|---|
| Routes of administration | Oral, smoked, inhaled |
| ATC code |
|
| Legal status | |
| Legal status |
|
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| ChemSpider | |
| UNII | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
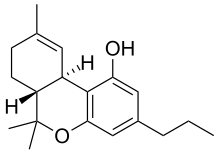
| Formula | C19H26O2 |
| Molar mass | 286.415 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
Tetrahydrocannabivarin (THCV, THV, O-4394, GWP42004) is a homologue of tetrahydrocannabinol (THC) having a propyl (3-carbon) side chain instead of pentyl (5-carbon), making it non-psychoactive in lower doses. It has been shown to exhibit neuroprotective activity, appetite suppression, glycemic control and reduced side effects compared to THC, making it a potential treatment for management of obesity and diabetes.[1] THCV was studied by Roger Adams as early as 1942.[2]
- ^ Abioye A, Ayodele O, Marinkovic A, Patidar R, Akinwekomi A, Sanyaolu A (January 2020). "Δ9-Tetrahydrocannabivarin (THCV): a commentary on potential therapeutic benefit for the management of obesity and diabetes". Journal of Cannabis Research. 2 (1): 6. doi:10.1186/s42238-020-0016-7. PMC 7819335. PMID 33526143.
- ^ Adams R, Loewe S, Smith CM, McPhee WD (1942). "Tetrahydrocannabinol Homologs and Analogs with Marihuana Activity. XIII1". Journal of the American Chemical Society. 64 (3): 694–697. doi:10.1021/ja01255a061.