| |

| |
| Names | |
|---|---|
IUPAC names
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ChemSpider | |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
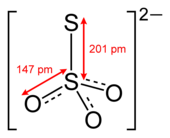
| S2O2−3 | |
| Molar mass | 112.12 g·mol−1 |
| Conjugate acid | Thiosulfuric acid |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Thiosulfate (IUPAC-recommended spelling; sometimes thiosulphate in British English) is an oxyanion of sulfur with the chemical formula S2O2−3. Thiosulfate also refers to the compounds containing this anion, which are the salts of thiosulfuric acid, such as sodium thiosulfate Na2S2O3 and ammonium thiosulfate (NH4)2S2O3. Thiosulfate salts occur naturally. Thiosulfate rapidly dechlorinates water, and is used to halt bleaching in the paper-making industry. Thiosulfate salts are mainly used for dyeing in textiles, and bleaching of natural substances.[2]
- ^ International Union of Pure and Applied Chemistry (2005). Nomenclature of Inorganic Chemistry (IUPAC Recommendations 2005). Cambridge (UK): RSC–IUPAC. ISBN 0-85404-438-8. pp. 139,329. Electronic version.
- ^ Cite error: The named reference
Ullmannwas invoked but never defined (see the help page).