| |
| |
| Names | |
|---|---|
| IUPAC name
Calcium phosphate
| |
| Other names
Tribasic calcium phosphate, tricalcium bis(phosphate)
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ChemSpider | |
| ECHA InfoCard | 100.028.946 |
| EC Number |
|
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
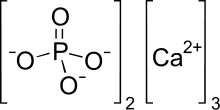
| Ca3(PO4)2 | |
| Molar mass | 310.18 g/mol |
| Appearance | White amorphous powder |
| Density | 3.14 g/cm3[1] |
| Melting point | 1,670 °C (3,040 °F; 1,940 K)[1] |
| 1.2 mg/kg[1] | |
Solubility product (Ksp)
|
2.07×10−33[2] |
| Thermochemistry | |
Std enthalpy of
formation (ΔfH⦵298) |
−4126 kJ/mol (α-form)[3] |
| Pharmacology | |
| A12AA01 (WHO) | |
| Hazards | |
| NFPA 704 (fire diamond) | |
| Flash point | Non-flammable |
| Related compounds | |
Other anions
|
Calcium pyrophosphate |
Other cations
|
Trimagnesium phosphate Trisodium phosphate Tripotassium phosphate |
Related compounds
|
Monocalcium phosphate Dicalcium phosphate |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Tricalcium phosphate (sometimes abbreviated TCP), more commonly known as Calcium phosphate, is a calcium salt of phosphoric acid with the chemical formula Ca3(PO4)2. It is also known as tribasic calcium phosphate and bone phosphate of lime (BPL). It is a white solid of low solubility. Most commercial samples of "tricalcium phosphate" are in fact hydroxyapatite.[4][5]
It exists as three crystalline polymorphs α, α′, and β. The α and α′ states are stable at high temperatures.
- ^ a b c Haynes, William M., ed. (2016). CRC Handbook of Chemistry and Physics (97th ed.). Boca Raton, FL: CRC Press. p. 4.54. ISBN 9781498754293.
- ^ John Rumble (June 18, 2018). CRC Handbook of Chemistry and Physics (99 ed.). CRC Press. pp. 5–188. ISBN 978-1-138-56163-2.
- ^ Zumdahl, Steven S. (2009). Chemical Principles 6th Ed. Houghton Mifflin Company. p. A21. ISBN 978-0-618-94690-7.
- ^ Klaus Schrödter; Gerhard Bettermann; Thomas Staffel; Friedrich Wahl; Thomas Klein; Thomas Hofmann (2012). "Phosphoric Acid and Phosphates". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a19_465.pub3. ISBN 978-3527306732.
- ^ El Moussaoui, Youssef; Terrisse, Hélène; Quillard, Sophie; Ropers, Marie-Hélène; Humbert, Bernard (January 2023). "The True Nature of Tricalcium Phosphate Used as Food Additive (E341(iii))". Nanomaterials. 13 (12): 1823. doi:10.3390/nano13121823. ISSN 2079-4991. PMC 10303396. PMID 37368253.
