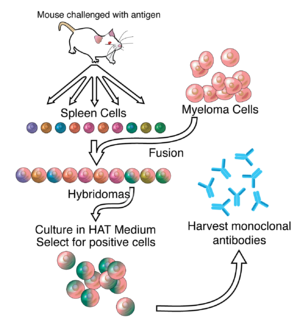
ZMapp is an experimental biopharmaceutical drug comprising three chimeric monoclonal antibodies under development as a treatment for Ebola virus disease.[1] Two of the three components were originally developed at the Public Health Agency of Canada's National Microbiology Laboratory (NML), and the third at the U.S. Army Medical Research Institute of Infectious Diseases; the cocktail was optimized by Gary Kobinger, a research scientist at the NML[2] and underwent further development under license by Mapp Biopharmaceutical. ZMapp was first used on humans during the Western African Ebola virus epidemic, having only been previously tested on animals and not yet subjected to a randomized controlled trial.[3] The National Institutes of Health (NIH) ran a clinical trial starting in January 2015 with subjects from Sierra Leone, Guinea, and Liberia aiming to enroll 200 people, but the epidemic waned and the trial closed early, leaving it too statistically underpowered to give a meaningful result about whether ZMapp worked.[4]
In 2016, a clinical study comparing ZMapp to the current standard of care for Ebola was inconclusive.[5]
- ^ a b Cite error: The named reference
Krollwas invoked but never defined (see the help page). - ^ Branswell, Helen (September 21, 2014). "How a Winnipeg lab became an Ebola research powerhouse". CBC News. The Canadian Press. Retrieved 23 November 2015.
- ^ Cite error: The named reference
NYTwhowas invoked but never defined (see the help page). - ^ "Press release: Study Finds Ebola Treatment ZMapp Holds Promise, Although Results Not Definitive | NIH: National Institute of Allergy and Infectious Diseases". National Institute of Allergy and Infectious Diseases. October 12, 2016.
- ^ PREVAIL II (October 13, 2016). "A Randomized, Controlled Trial of ZMapp for Ebola Virus Infection". NEJM. 375 (15): 1448–1456. doi:10.1056/NEJMoa1604330. PMC 5086427. PMID 27732819.