| |

| |
| Names | |
|---|---|
| Other names
Zirconium(IV) carbide
| |
| Identifiers | |
3D model (JSmol)
|
|
| ECHA InfoCard | 100.031.920 |
| EC Number |
|
PubChem CID
|
|
| RTECS number |
|
| UN number | 3178 |
CompTox Dashboard (EPA)
|
|
| |
| Properties | |
| ZrC | |
| Molar mass | 103.235 g·mol−1 |
| Appearance | Gray refractory solid |
| Odor | Odorless |
| Density | 6.73 g/cm3 (24 °C)[1] |
| Melting point | 3,532–3,540 °C (6,390–6,404 °F; 3,805–3,813 K)[1][2] |
| Boiling point | 5,100 °C (9,210 °F; 5,370 K)[2] |
| Insoluble | |
| Solubility | Soluble in concentrated H2SO4, HF,[1] HNO3 |
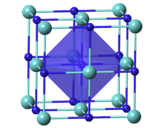
| Structure | |
| Cubic, cF8[3] | |
| Fm3m, No. 225[3] | |
a = 4.6976(4) Å[3] α = 90°, β = 90°, γ = 90°
| |
| Octahedral[3] | |
| Thermochemistry | |
Heat capacity (C)
|
37.442 J/mol·K[4] |
Std molar
entropy (S⦵298) |
33.14 J/mol·K[4] |
Std enthalpy of
formation (ΔfH⦵298) |
−207 kJ/mol (extrapolated to stoichiometric composition)[5] −196.65 kJ/mol[4] |
| Hazards | |
| Occupational safety and health (OHS/OSH): | |
Main hazards
|
Pyrophoric |
| GHS labelling: | |
  [6] [6]
| |
| Danger | |
| H228, H302, H312, H332[6] | |
| P210, P280[6] | |
| NFPA 704 (fire diamond) | |
| Related compounds | |
Other anions
|
Zirconium nitride Zirconium oxide |
Other cations
|
Titanium carbide Hafnium carbide Vanadium carbide Niobium carbide Tantalum carbide Chromium carbide Molybdenum carbide Tungsten carbide |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Zirconium carbide (ZrC) is an extremely hard refractory ceramic material,[7] commercially used in tool bits for cutting tools. It is usually processed by sintering.
- ^ a b c Lide, David R., ed. (2009). CRC Handbook of Chemistry and Physics (90th ed.). Boca Raton, Florida: CRC Press. ISBN 978-1-4200-9084-0.
- ^ a b Perry, Dale L. (2011). Handbook of Inorganic Compounds (2nd ed.). CRC Press. p. 472. ISBN 978-1-4398-1461-1.
- ^ a b c d Kempter, C. P.; Fries, R. J. (1960). "Crystallographic Data. 189. Zirconium Carbide". Analytical Chemistry. 32 (4): 570. doi:10.1021/ac60160a042.
- ^ a b c Zirconium carbide in Linstrom, Peter J.; Mallard, William G. (eds.); NIST Chemistry WebBook, NIST Standard Reference Database Number 69, National Institute of Standards and Technology, Gaithersburg (MD) (retrieved 2014-06-30)
- ^ Baker, F. B.; Storms, E. K.; Holley, C. E. (1969). "Enthalpy of formation of zirconium carbide". Journal of Chemical & Engineering Data. 14 (2): 244. doi:10.1021/je60041a034.
- ^ a b c Sigma-Aldrich Co., Zirconium(IV) carbide. Retrieved on 2014-06-30.
- ^ Measurement and theory of the hardness of transition- metal carbides , especially tantalum carbide. Schwab, G. M.; Krebs, A. Phys.-Chem. Inst., Univ. Muenchen, Munich, Fed. Rep. Ger. Planseeberichte fuer Pulvermetallurgie (1971), 19(2), 91-110
