 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Imovane, Zimovane, Dopareel, others |
| AHFS/Drugs.com | International Drug Names |
| Pregnancy category |
|
| Routes of administration | Oral tablets, 3.75 mg or 7.5mg (UK), 5 mg, 7.5 mg, or 10 mg (JP) |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | 75–80%[2] |
| Protein binding | 52–59% |
| Metabolism | Hepatic through CYP3A4 and CYP2E1 |
| Elimination half-life | ~5 hours (3.5–6.5 hours) ~7–9 hours for 65+ years old |
| Excretion | Urine (80%) |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| PDB ligand | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.051.018 |
| Chemical and physical data | |
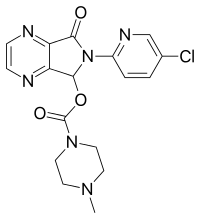
| Formula | C17H17ClN6O3 |
| Molar mass | 388.81 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| (verify) | |
Zopiclone, sold under the brand name Imovane among others, is a nonbenzodiazepine, specifically a cyclopyrrolone, used to treat difficulty sleeping. Zopiclone is molecularly distinct from benzodiazepine drugs and is classed as a cyclopyrrolone. However, zopiclone increases the normal transmission of the neurotransmitter gamma-aminobutyric acid (GABA) in the central nervous system, via modulating GABAA receptors similarly to the way benzodiazepine drugs do inducing sedation but not with the anti-anxiety properties of the benzodiazepines.
Zopiclone is a sedative. It works by causing a depression or tranquilization of the central nervous system. After prolonged use, the body can become accustomed to the effects of zopiclone. When the dose is then reduced or the drug is abruptly stopped, withdrawal symptoms may result. These can include a range of symptoms similar to those of benzodiazepine withdrawal. Although withdrawal symptoms from therapeutic doses of zopiclone and its isomers (i.e., eszopiclone) do not typically present with convulsions and are therefore not considered life-threatening, patients may experience such significant agitation or anxiety that they seek emergency medical attention. [citation needed]
In the United States, zopiclone is not commercially available,[3] although its active stereoisomer, eszopiclone, is. Zopiclone is a controlled substance in the United States, Japan, Brazil, New Zealand and some European countries, and may be illegal to possess without a prescription. [citation needed]
Zopiclone is known colloquially as a "Z-drug". Other Z-drugs include zaleplon and zolpidem and were initially thought to be less addictive than benzodiazepines. However, this appraisal has shifted somewhat in the last few years as cases of addiction and habituation have been presented. Zopiclone is recommended to be taken at the lowest effective dose, with a duration of 2–3 weeks for short-term insomnia.[4] Daily or continuous use of the drug is not usually advised, and caution must be taken when the compound is used in conjunction with benzodiazepines, sedatives or other drugs affecting the central nervous system.[5]
- ^ Anvisa (2023-03-31). "RDC Nº 784 - Listas de Substâncias Entorpecentes, Psicotrópicas, Precursoras e Outras sob Controle Especial" [Collegiate Board Resolution No. 784 - Lists of Narcotic, Psychotropic, Precursor, and Other Substances under Special Control] (in Brazilian Portuguese). Diário Oficial da União (published 2023-04-04). Archived from the original on 2023-08-03. Retrieved 2023-08-16.
- ^ "Assessment of Zopiclone" (PDF). World Health Organization. Essential Medicines and Health Products. World Health Organization. 2006. p. 9 (Section 5. Pharmacokinetics). Retrieved 5 December 2015.
- ^ "Zopiclone consumer information from". Drugs.com. Retrieved 2013-06-06.
- ^ "Chapter 3 - The technologies, section 3.4", Clinical need and practice - Guidance on the use of zaleplon, zolpidem and zopiclone for the short-term management of insomnia, National Institute for Health and Care Excellence (NICE), 28 April 2004, Technology appraisal guidance [TA77],
This guidance will be reviewed if there is new evidence.
Current as of 8 June 2023 - ^ Van Der Kleijn E (1989). "Effects of zopiclone and temazepam on sleep, behaviour and mood during the day". European Journal of Clinical Pharmacology. 36 (3): 247–251. doi:10.1007/BF00558155. ISSN 0031-6970. PMID 2744064.